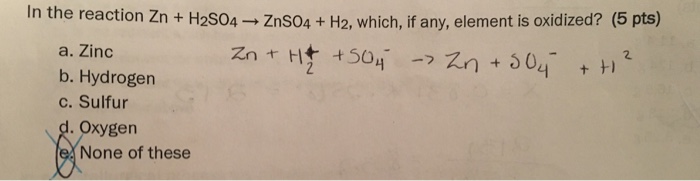Question Video: Identifying the Name of Ions that Are Not Included in the Net Ionic Equation | Nagwa
Consider the following reactions: I. Zn + Dil. H2SO4 → ZnSO4 + H2 II. Zn + Conc. H2SO4 → ZnSO4 + SO2 + H2O - Sarthaks eConnect | Largest Online Education Community
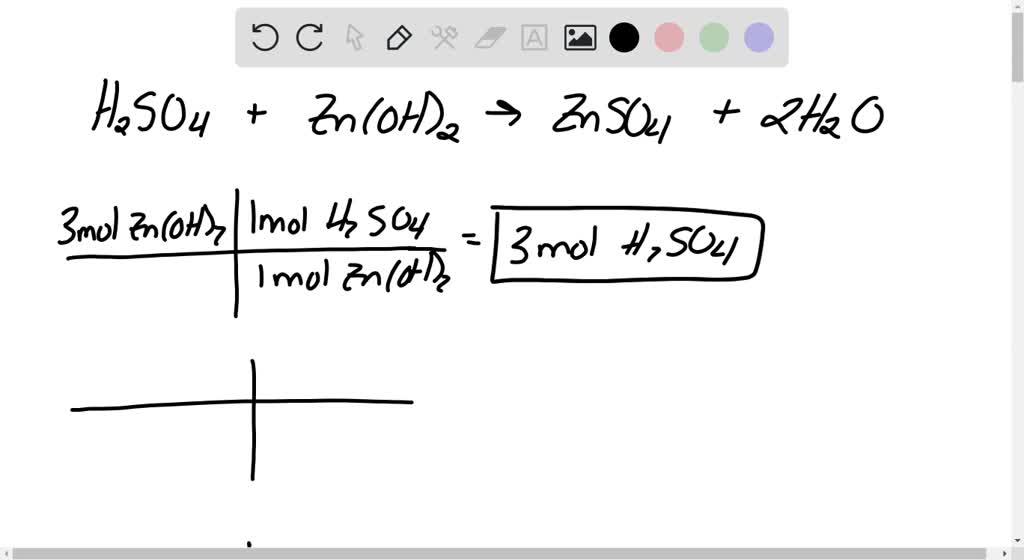
SOLVED: When sulfuric acid reacts with zinc hydroxide, zinc sulfate and water are produced. The balanced equation for this reaction is: H2SO4 (aq) + Zn(OH)2 (s) –> ZnSO4 (aq) + 2H2O (l)

SOLVED: The balanced chemical equation for the reaction between sulfuric acid and zinc hydroride is: HzSO ( aq )- Zn(OH) ( ) ZnSO4 ( aq ) + 2 Hzo ( 4) We

Question Video: Identifying the Symbol Equation That Represents the Reaction of Zinc with Sulfuric Acid | Nagwa

How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) | How to Balance H2SO4 + Zn = ZnSO4 + H2 (Sulfuric Acid + Zinc) Balancing equations could